| Results of a recent “proof of concept” trial on mild cognitive decline and early Alzheimer’s disease was published in May this year in medRxiv.org. Currently the medical model of treating Alzheimer’s and mild cognitive impairment is the single drug approach which has not been found to be effective. In the trial 25 patients with Alzheimer’s disease or mild cognitive impairment aged 50 to 76 years participated in the trial. In the group were 13 women and 12 men. All patients were evaluated with a series of tests including pathology testing, cognitive assessments, gene testing and functional medicine tests. |
The patients were all given an MRI of the brain including volumetrics (something that is not yet widely included in Australia). Each patient undertook a CNS Vital Signs online cognitive assessment and a Montreal Cognitive Assessment (MoCA). A family member or partner also completed a questionnaire regarding the patient’s cognitive function.
The patients were then treated for nine months with a personalised protocol to address the patient’s specific contributing factors. (The protocol used was the Bredesen ReCode protocol.) Cognitive function was assessed every three months. The treatment included dietary improvements, nutritional and herbal supplements and hormone treatment where indicated to address deficiencies. The treatment was aimed at targeting the drivers present such as inflammation, insulin and glucose metabolism issues, toxicity, and pathogen infections. Further consideration was given to sleep, exercise, and stress management and brain training during the treatment.
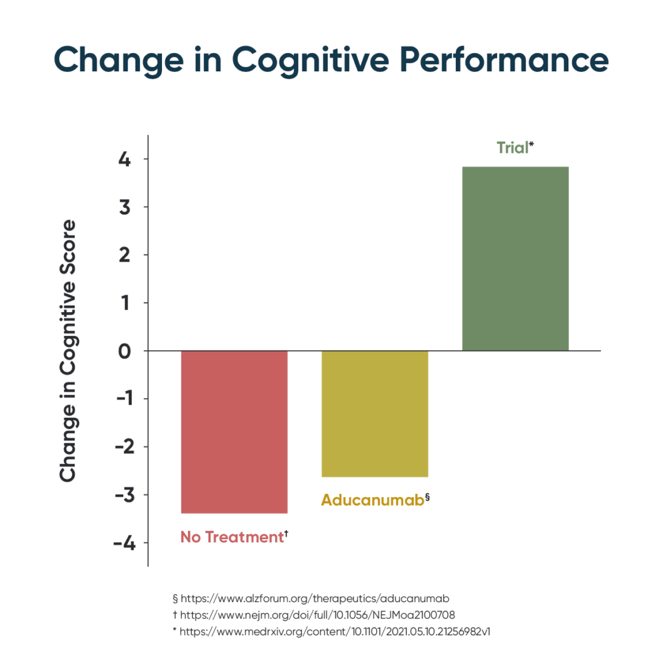
Twenty-one of the 25 patients improved in their CNS Vital signs neurocognitive index scores at each three-month test. Eighty four percent of patients improved over the nine months, 12 percent had a decline in their cognitive function and four percent had no change.
It was concluded that the results support the view that determining the underlying contributors to the cognitive decline for each patient and then treating with a personalised protocol represents an effective treatment approach for patients with MCI or early dementia.
This proof of concept has proven that a larger, randomised controlled trial is warranted. There is already a follow up study in the works. Watch this space!
You can read more about the Bredesen Recode Protocol here.
The patients were then treated for nine months with a personalised protocol to address the patient’s specific contributing factors. (The protocol used was the Bredesen ReCode protocol.) Cognitive function was assessed every three months. The treatment included dietary improvements, nutritional and herbal supplements and hormone treatment where indicated to address deficiencies. The treatment was aimed at targeting the drivers present such as inflammation, insulin and glucose metabolism issues, toxicity, and pathogen infections. Further consideration was given to sleep, exercise, and stress management and brain training during the treatment.
Twenty-one of the 25 patients improved in their CNS Vital signs neurocognitive index scores at each three-month test. Eighty four percent of patients improved over the nine months, 12 percent had a decline in their cognitive function and four percent had no change.
It was concluded that the results support the view that determining the underlying contributors to the cognitive decline for each patient and then treating with a personalised protocol represents an effective treatment approach for patients with MCI or early dementia.
This proof of concept has proven that a larger, randomised controlled trial is warranted. There is already a follow up study in the works. Watch this space!
You can read more about the Bredesen Recode Protocol here.

 RSS Feed
RSS Feed